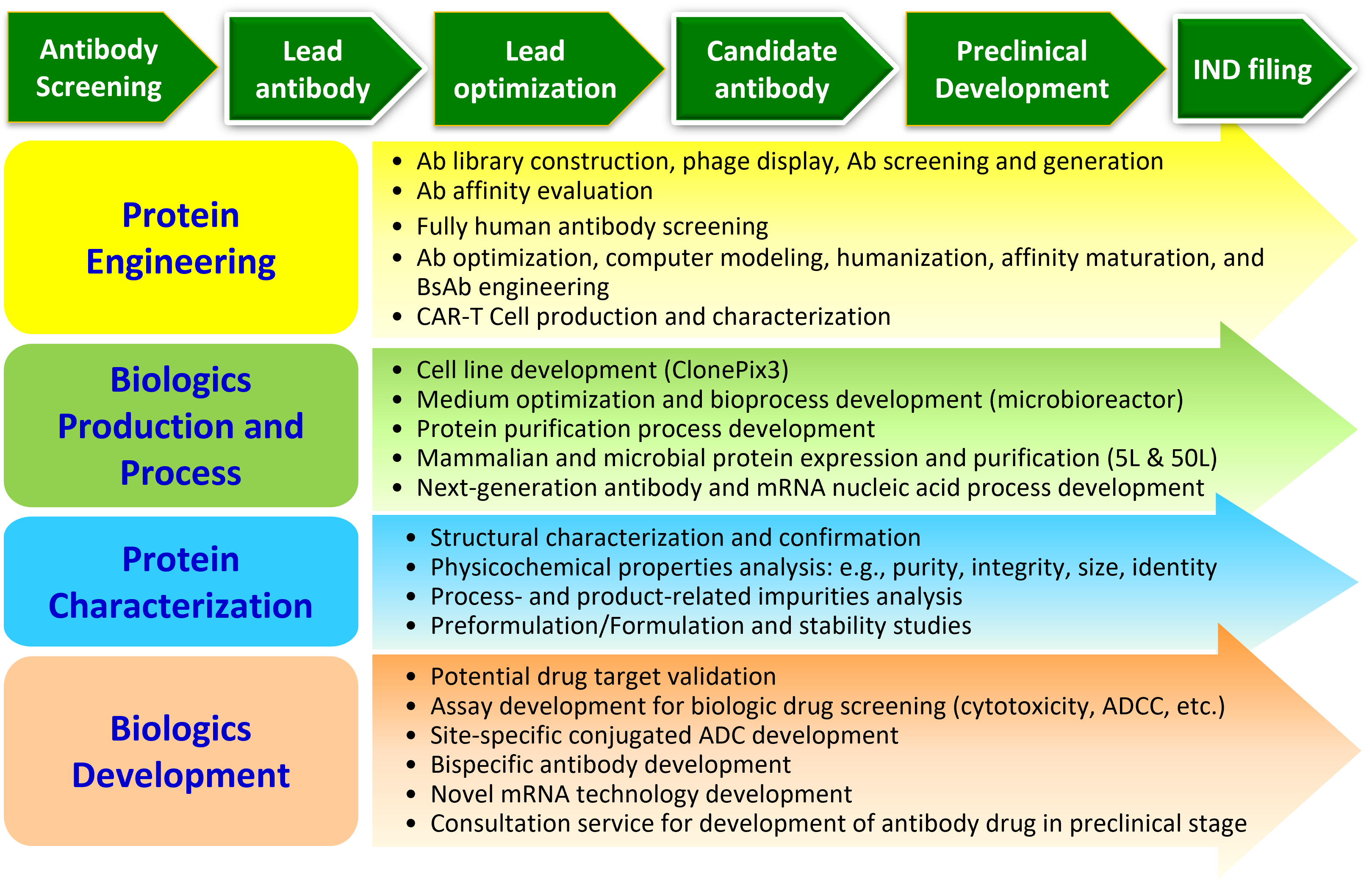
DCB's Institute of Biologics is devoted to developing therapeutic antibodies and next generation biologics including bispecific antibodies (BsAb) and antibody-drug conjugates (ADC). Our integrated capabilities are from library construction and screening, humanization, affinity maturation, cell line development, pharmacology, and PK/PD to toxicology of antibody development. With the help of these capabilities, the institute has created a number of unique biologics candidates and some of them are under clinical development in combination with local cGMP manufacture industries. We recently also focus on the establishment of novel immune cell therapy technology platforms and mRNA technology platforms including chimeric antigen receptor T cells (CAR-T), self-amplifying mRNA and circular RNA.
In addition to new drug development, the Institute of Biologics also provides services to the academic community and biotechnology industry. These services include antibody screening and engineering, cell line development, protein expression, process development, functional assays, protein characterization and cell line engineering.
>>>Download leaflets of the Institute of Biologics